|
Product Information |
|
Product name |
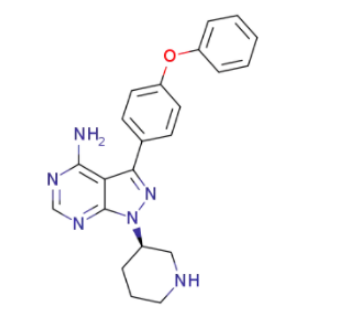
3-(4-phenoxyphenyl)-1-(3-piperidyl)pyrazolo[3,4-d]pyrimidin-4-amine;Ibrutinib intermediate |
|
CAS No. |
1022150-12-4 |
|
Molecular Formula |
C22H22N6O |
|
Molecular Weight |
386.456 |
|
Molecular Structure |
 |
|
Quality Standard |
99% up by HPLC |
|
Appearance |
White to off-white powder |